Meet the Omori UK team at Interpack 2026
Whether you’re exploring new equipment, planning upgrades, or tackling ongoing production challenges, our team will be on hand to help.

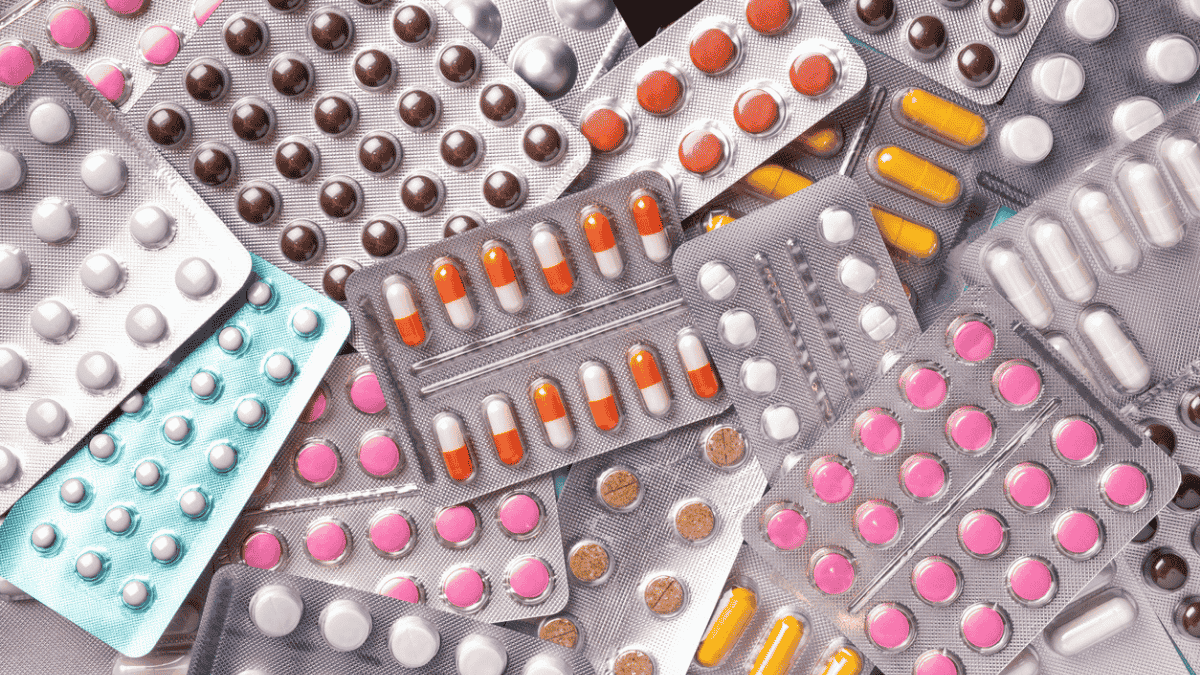
Packaging plays a crucial role in the pharmaceutical industry, ensuring that medicines remain effective, safe and easy to handle. The packaging process is categorised into three levels: primary, secondary and tertiary packaging. Each serves a specific function in protecting pharmaceutical products throughout their lifecycle.
In this guide, we’ll explore the difference between primary and secondary packaging in pharmaceuticals, detailing their key roles, materials used and use cases in the industry.
Primary packaging is the first layer of protection that comes into direct contact with the pharmaceutical product. Its primary function is to preserve product integrity, prevent contamination and ensure the correct dosage is delivered to the end-user.
The choice of primary packaging materials for pharmaceuticals depends on the nature of the drug and the required storage conditions. Common materials include:
Some common primary packaging examples in the pharmaceutical industry include:
Secondary packaging in the pharmaceutical industry is the external layer that holds and protects primary packaging units. While it does not come into direct contact with the drug, its role is critical in branding, safety and logistics.
| Feature | Primary Packaging | Secondary Packaging |
| Direct contact with drug | Yes | No |
| Function | Protects, preserves, and maintains product sterility | Groups, protects, and provides branding & information |
| Examples | Blister packs, vials, syringes, ampoules | Cartons, boxes, shrink wraps |
| Regulatory compliance | Must meet strict pharmaceutical packaging regulations | Ensures safety, branding, and handling efficiency |
| Materials used | Glass, plastic, rubber, aluminium foil | Cardboard, paper, plastic films |
Understanding these differences is vital in ensuring that pharmaceutical products reach consumers safely and in compliance with regulatory standards.
Pharmaceutical packaging regulations set strict guidelines on how medicines should be packaged to ensure patient safety. Secondary packaging plays a crucial role in meeting these requirements by including:
Secondary packaging ensures that:
Properly designed secondary packaging optimises transportation and storage, reducing the risk of damage and making inventory management more efficient.
While primary and secondary packaging serve crucial roles in product protection and information, tertiary packaging ensures the safe and efficient transport of pharmaceutical products on a larger scale. It is designed to safeguard large quantities of medicine during bulk transportation, handling and storage, preventing physical damage while maintaining the integrity of the drugs inside.
Tertiary packaging is not consumer-facing and is primarily used for logistics and warehouse storage, ensuring that pharmaceuticals reach pharmacies, hospitals and distributors safely. This layer of packaging plays a significant role in supply chain efficiency, sustainability and cost-effectiveness, as it optimises space and enhances transportation security.
Tertiary packaging may not be as visible as primary or secondary packaging, but it is a critical component in maintaining the quality and safety of pharmaceutical products throughout the entire supply chain.
As the pharmaceutical industry continues to evolve, innovations in secondary packaging are becoming increasingly crucial for product safety, regulatory compliance and efficiency. New developments in pharmaceutical packaging machinery are improving security, sustainability and patient engagement, addressing many challenges faced by the industry today.
One of the most significant advancements in pharmaceutical packaging is smart packaging technology, which integrates digital elements such as QR codes, NFC (Near Field Communication) and RFID (Radio Frequency Identification) tags. These features enable:
With increasing global emphasis on sustainability, pharmaceutical companies are focusing on reducing waste and using environmentally friendly packaging materials. Key innovations include:
Counterfeit medicines are a significant global concern, putting patient safety at risk and damaging brand reputations. Innovative anti-counterfeiting technologies are being integrated into secondary packaging for pharmaceutical products to prevent unauthorised tampering and forgery. These include:
Pharmaceutical secondary packaging is going through major shifts in direction, with innovations focusing on safety, efficiency and sustainability. These advancements not only improve product protection but also enhance patient trust and regulatory compliance, ensuring that medicines remain safe, effective and accessible.
The role of primary, secondary and tertiary packaging in pharmaceuticals is essential for maintaining product integrity, compliance and patient safety. While primary packaging ensures direct protection of the drug, secondary packaging plays a vital role in branding, security and distribution efficiency. Meanwhile, tertiary packaging guarantees the safe transportation and bulk storage of pharmaceutical products, helping pharmaceutical manufacturers maintain a seamless and secure supply chain.
At Omori, we specialise in pharmaceutical packaging solutions tailored to meet regulatory and industry standards. If you’re looking for reliable and innovative pharmaceutical packaging machinery, explore our solutions to ensure efficiency and compliance in your packaging processes.
Interested in our packaging solutions, have a question for our team or want to arrange an equipment trial?
Our team is always happy to answer any questions you may have and provide you with the information you need to make the right decision for your business.
Contact us today and one of our experts will get back to you.